💡
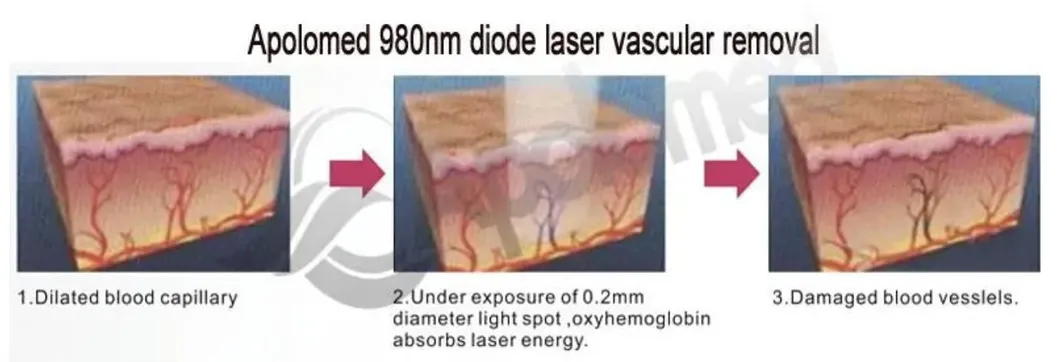
Working Principle
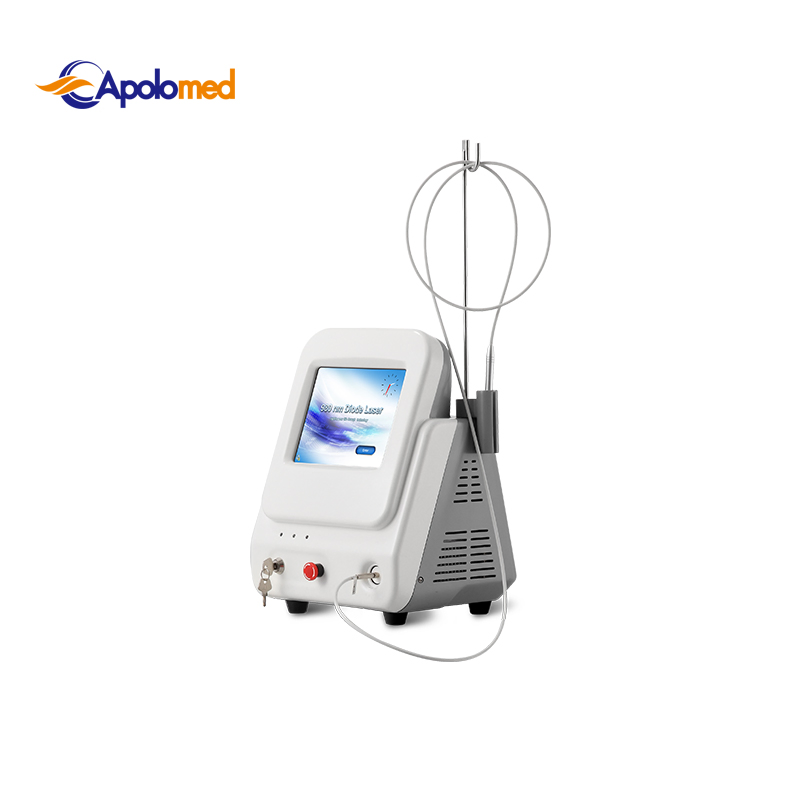
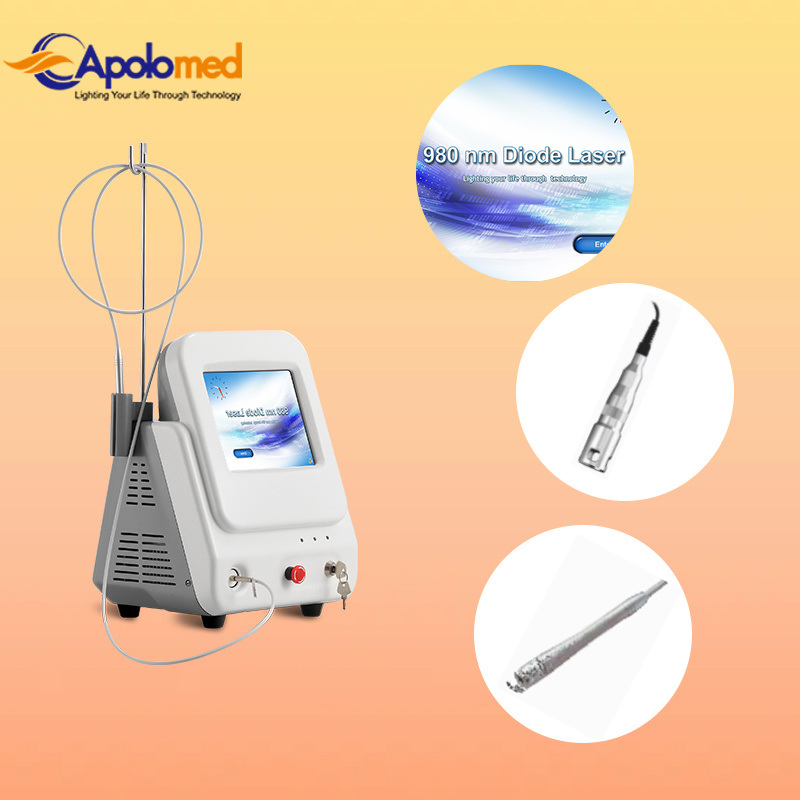
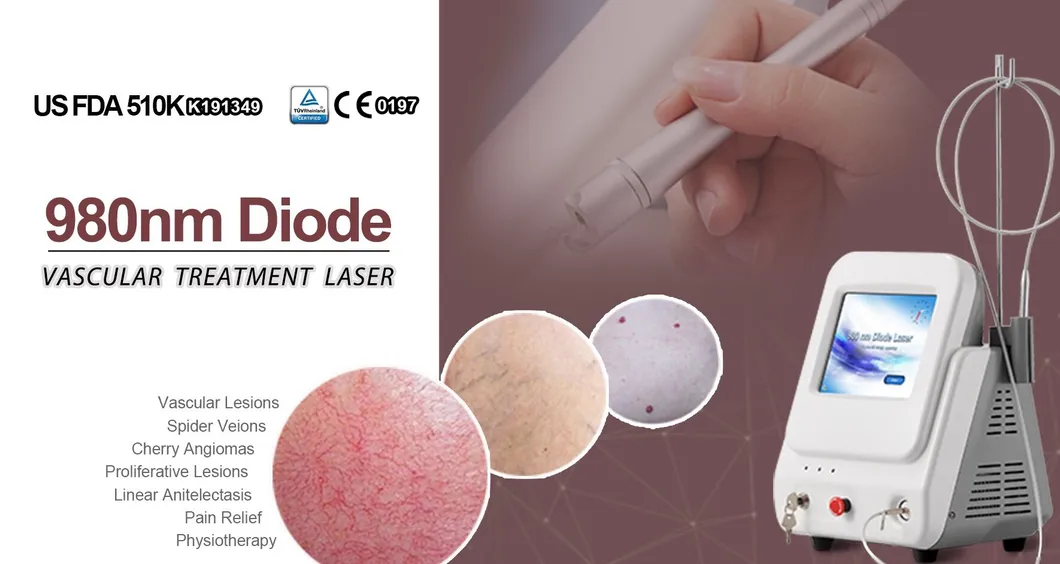
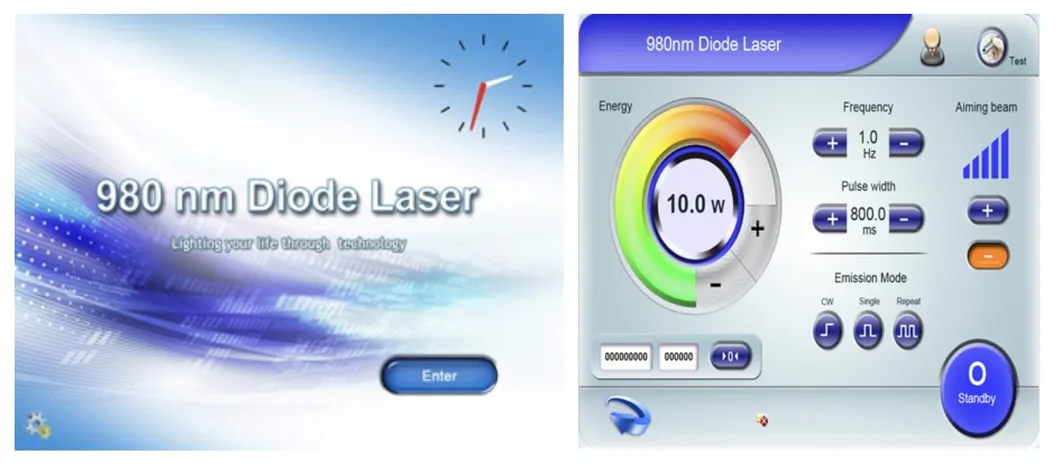
Based on the theory of "selective laser photothermal", the 980nm diode laser system uses specific wavelength to penetrate into skin for vascular treatment. Under laser irradiation, hemoglobin and red pigment capillaries maximize absorption of laser energy, causing solidification, blocking blood vessels, and leading to intravascular coagulation and final metabolic degradation.
🏢
Company Capability
As a leading designer and manufacturer since 2001, we specialize in IPL, Various Laser technologies, HIFU, and Body Slimming solutions. We manufacture equipment strictly in accordance with ISO 13485 and all products comply with Medical CE certificates (MDD & MDR).
❓
Frequently Asked Questions
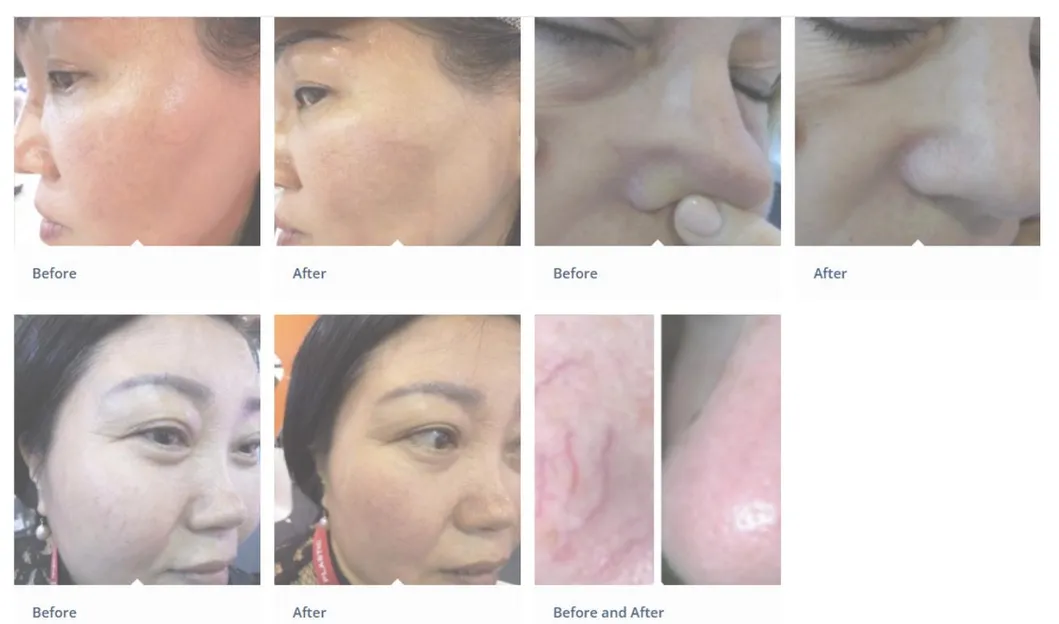
What is the main application of the HS-890 device?
It is primarily used for vascular lesion removal, including spider veins, cherry angiomas, and other blood vessel-related skin concerns on both the face and body.
Is the treatment painful or requires downtime?
The 980nm laser is highly selective, targeting only the hemoglobin. Most patients experience minimal discomfort, and there is typically no downtime, allowing for immediate return to daily activities.
What certifications does this laser machine hold?
The device is manufactured under ISO 13485 standards and holds Medical CE (MDR) certification and USA FDA 510K clearance.
How long does a typical treatment session take?
Depending on the area being treated, a session usually lasts between 30 to 45 minutes.
Can the machine be easily transported?
Yes, it features a portable design with a small footprint and weighs only 8kg, making it ideal for clinics with limited space or multi-room use.
Does the machine support OEM/ODM services?
Yes, as a professional designer and manufacturer, we provide full OEM and ODM support for medical and aesthetic devices.