










| Model | HS-890 |
|---|---|
| Wavelength | 980nm |
| Application | Commercial / Medical / Aesthetic |
| Output Power | 2~30W |
| Output Modes | CW, Single or Repeat Pulse |
| Pulse Width | 5~400ms |
| Certification | CE0197, ISO13485, FDA 510K |
| Cooling System | Air Cooling |
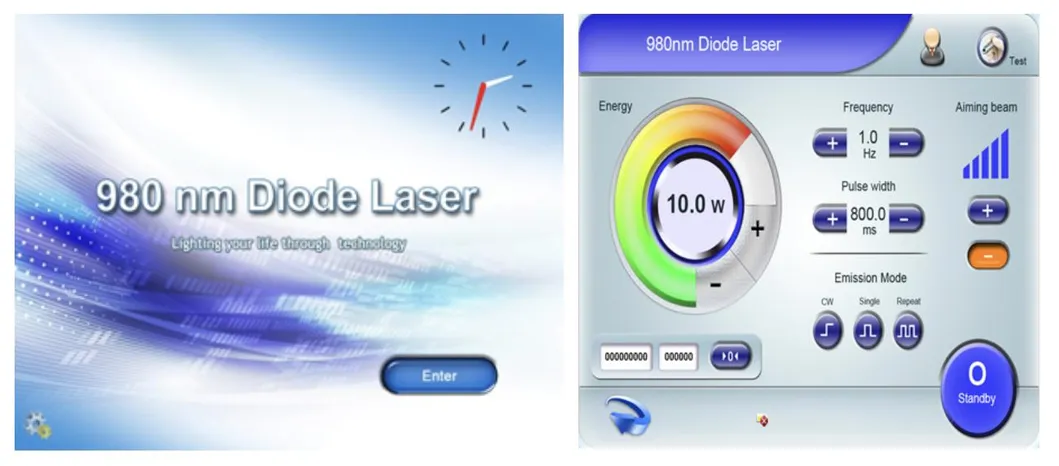
| Control Mode | 8" True Color Touch Screen |
| Dimensions | 28*27*37cm (L*W*H) |
| Weight | 8Kgs |
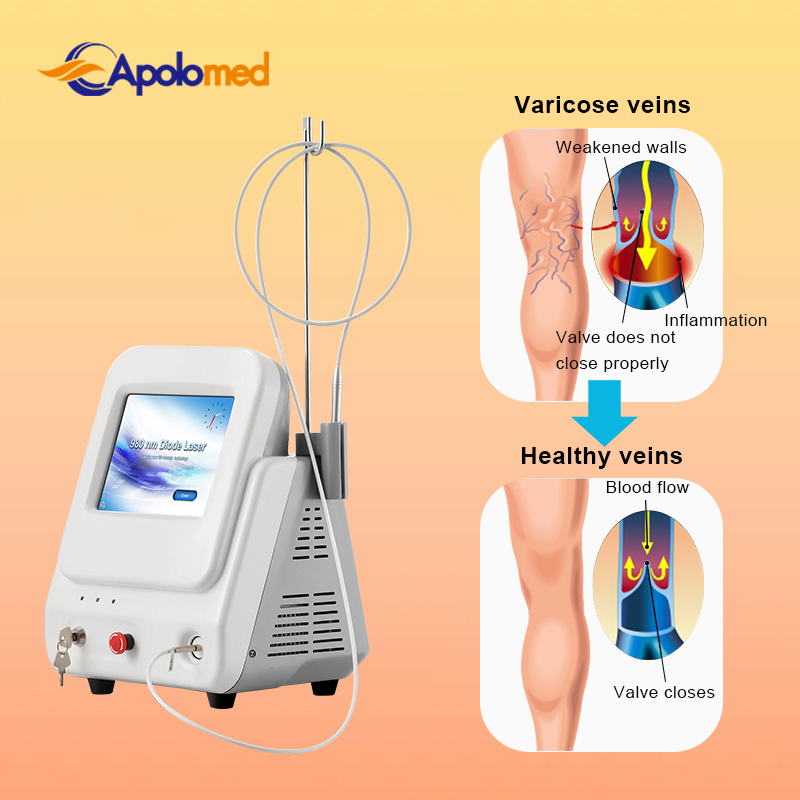
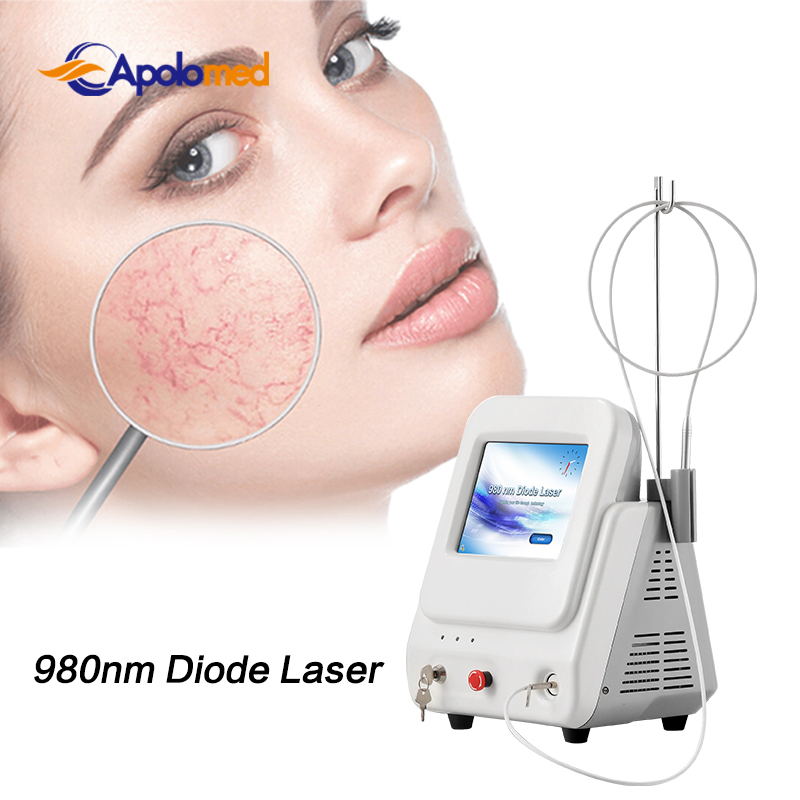
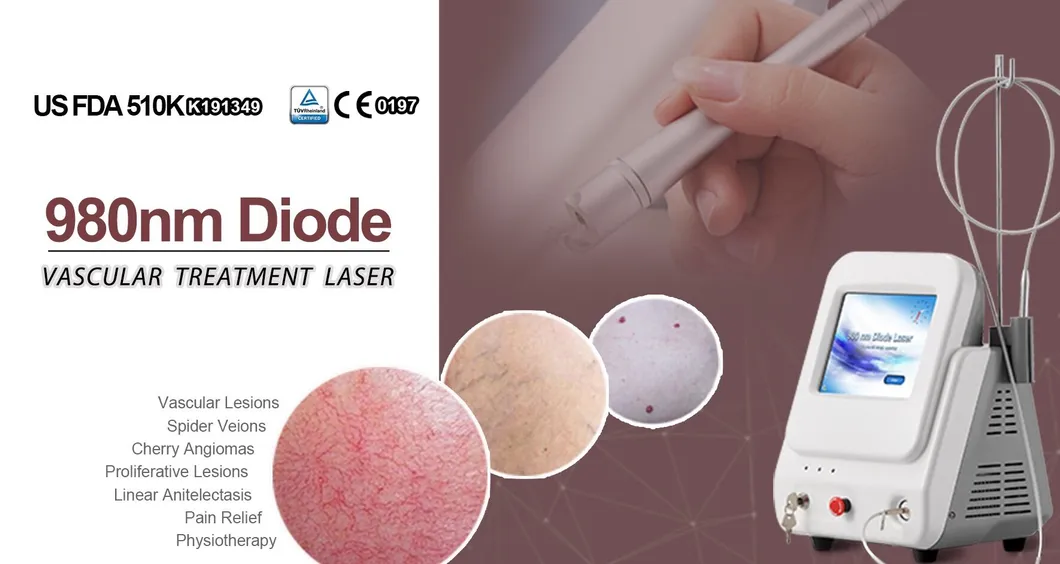
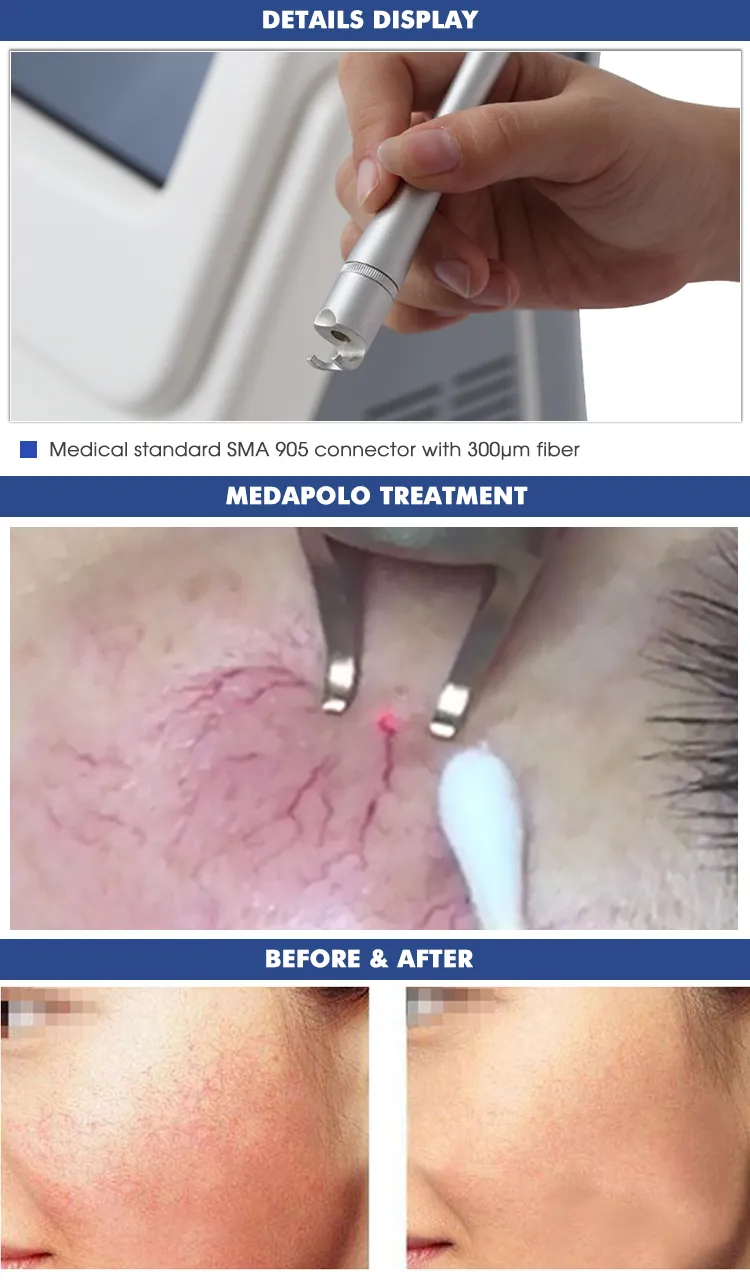
Based on the theory of "selective laser photothermal", the 980nm diode laser system uses specific wavelength energy to penetrate skin for vascular treatment. During irradiation, hemoglobin and red pigments in capillaries maximize absorption of laser energy, causing solidification and blocking of blood vessels. This leads to intravascular coagulation and final metabolic degradation.
The 980nm wavelength ensures that the normal skin architecture remains intact while providing a superior therapeutic effect without damaging superficial layers.

| Wavelength | 980nm |
|---|---|
| Laser Output Power | 2~30W |
| Output Modes | CW, Single or Repeat Pulse |
| Pulse Width | 5, 10~400ms |
| Pulse Repetition Rate | 1, 2, 3, 5, 10~50Hz |
| Single Pulse Energy | 0.1~12J |
| Transmission System | Fibers of 300um with SMAS 905 connector |
| Aiming Beam | Diode 650nm (red), ≤2mW |
| Power Rating | AC 100V or 240V, 50/60Hz |
Our facility is a leading designer and manufacturer of Intense Pulsed Light (IPL), Various Laser technologies, HIFU, and Body Slimming systems. Since 2001, we have developed over 40 high-standard devices with patented technologies to meet global aesthetic requirements.
We manufacture equipment strictly in accordance with ISO 13485 and all products comply with Medical CE certificates and US FDA 510K regulations.




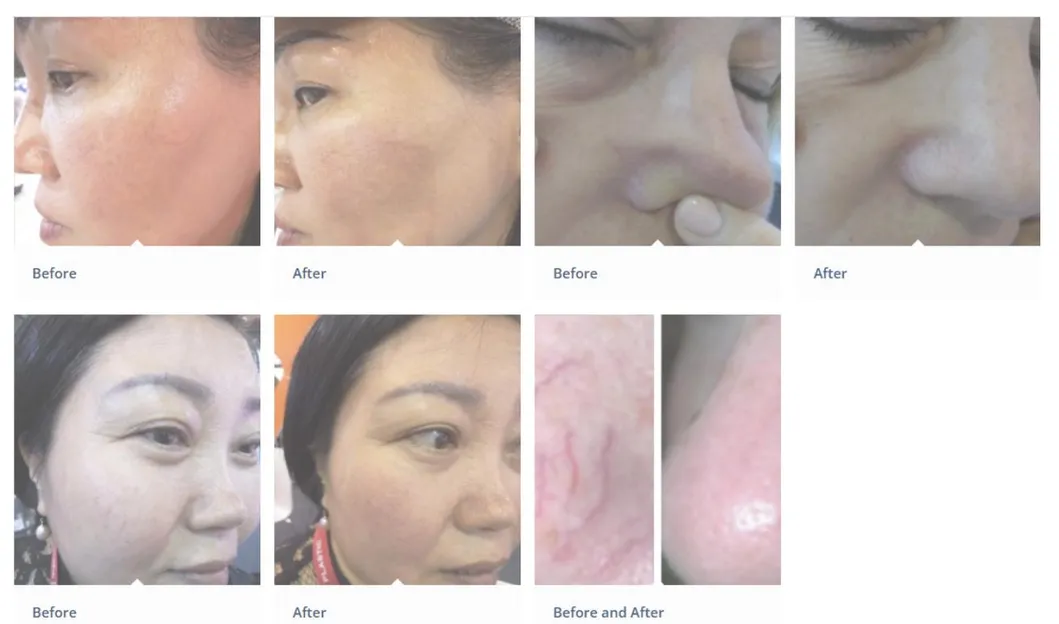
A: It is primarily used for vascular lesion removal, including spider veins on the face and body, cherry angiomas, and other blood vessel-related skin concerns.
A: The 980nm wavelength is highly selective, minimizing damage to surrounding tissue. Most procedures take 30-45 minutes with no significant downtime required.
A: The system is Medical CE (TUV) approved, ISO 13485 certified, and has cleared USA FDA 510K requirements.
A: It is designed for portability, weighing only 8kgs with a compact footprint of 28*27*37cm, making it ideal for various clinical settings.
A: Yes, the system features adjustable pulse widths from 5ms to 400ms and power levels from 2W to 30W to suit different treatment needs.
A: It utilizes an efficient air cooling system, which keeps the device compact while maintaining stable operation during commercial use.